Success Story
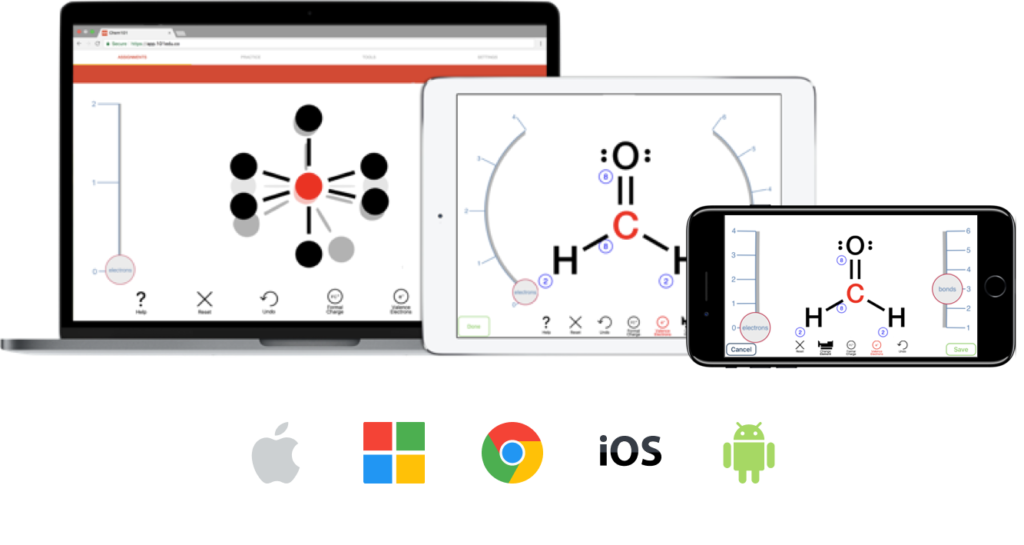
Professors from over 700 colleges and universities use Aktiv Chemistry to engage students inside and outside of the classroom. Learn how some of them have transformed their courses.


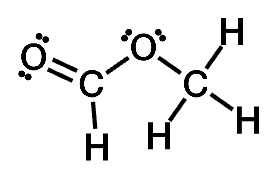
Helping Students Learn, Interact, and Visualize Chemistry in New Ways
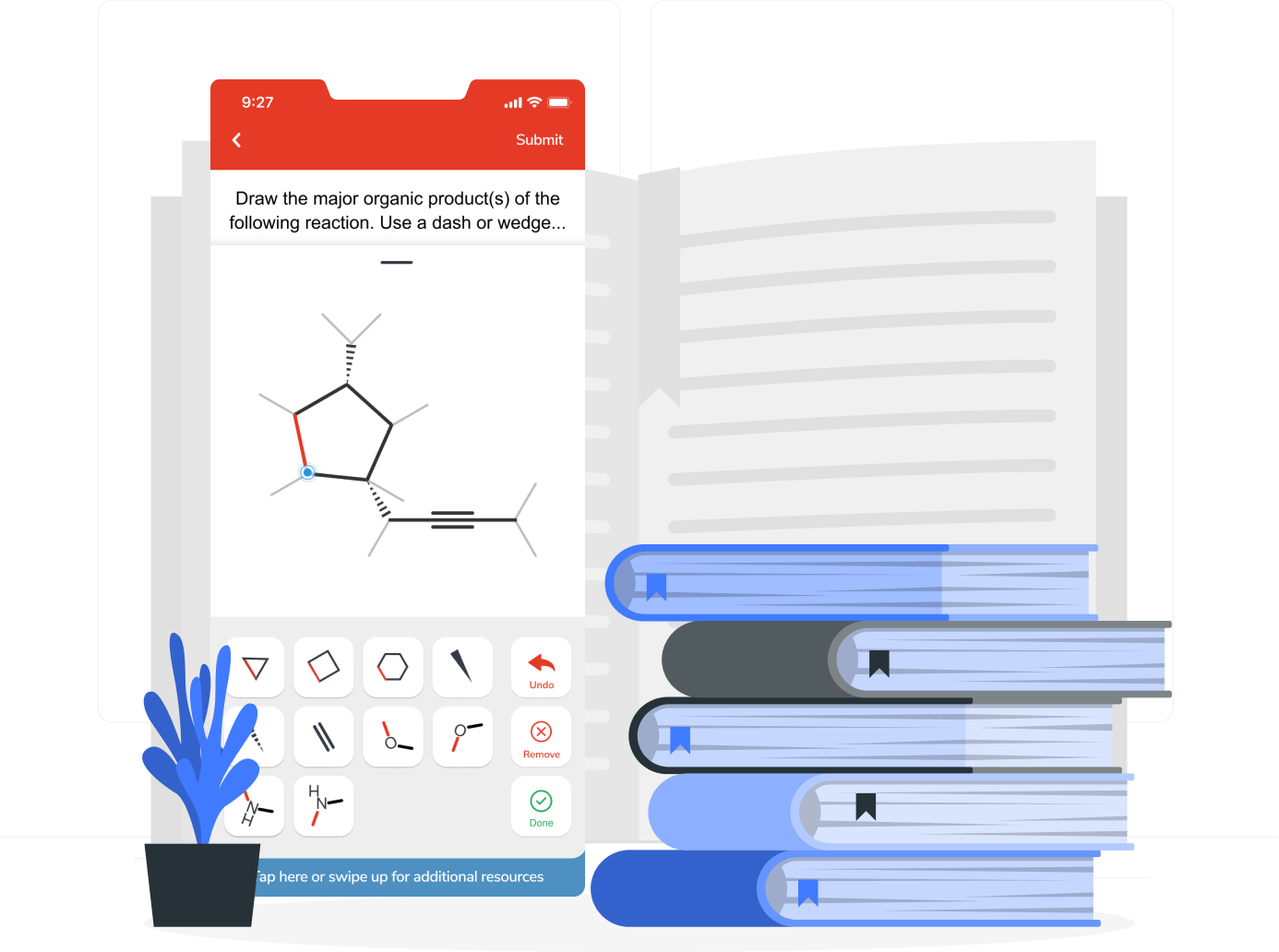
“With Aktiv Chemistry, there has been no learning curve. The students come in with a minimal amount of instruction and have been up and running within five to ten minutes. Then, they can focus more on the content rather than how to use the tool.”

Kathleen Kolbet
Professor of Chemistry, Department of Physical Sciences, Truckee Meadows Community College
¹ This quote was provided at a time where Aktiv Chemistry was named Chem101. We have replaced the use of Chem101 in any direct quotes with Aktiv Chemistry to minimize confusion.